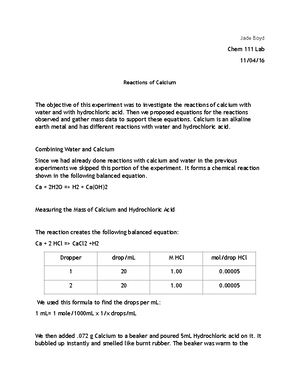
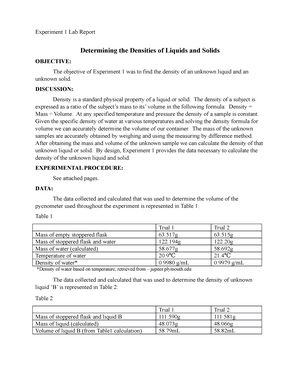
Which subatomic particles have approximately equal masses a
Anonymous Student
Subject:Chemistry
Which subatomic particles have approximately equal masses? a. protons and neutrons b. protons and electrons c. all protons d. neutrons and electrons e. none of the above
Answer
The correct answer is option a. The subatomic particles having approximately similar masses are neutrons and protons and the values of their masses respectively are 1.6749✕10-27 kg
- Discover more from:
Related Answered Questions
- General Chemistry I (CHEM 111)
Which of the following is not true regarding cations and anions? a. N3- represents a nitrogen atom that has gained three electrons b. An oxygen molecule can lose electron to form O2 - c. Ca2+ represents a calcium atom that has lost two electrons d. The formation of a chlorine anion can be written as Cl + e- ® Cle. N2 ® N2+ + e
Answers - General Chemistry I (CHEM 111)
Which subatomic particles have opposite charges? a. protons and neutrons b. protons and electrons c. all protons d. neutrons and electrons e. all neutrons
Answers - General Chemistry I (CHEM 111)
Which statement regarding Rutherford’s nuclear model of the atom is false? a. The diameter of the atom is approximately 10, 000 times larger than the nucleus b. The nucleus is approximately 100 times smaller than the diameter of the atom c. The nucleus is surrounded by a diffuse cloud of electrons d. Electrons and protons are not mix
Answers - General Chemistry I (CHEM 111)
Isotopes have_______________ a. the atomic mass b. the same total number of protons and neutrons c. the same number of protons but a different number of neutrons d. the same number of neutrons but a different number of protons e. the same number of protons but a different number of electrons
Answers - General Chemistry I (CHEM 111)
Which statement regarding Millikan’s oil drop experiment is true? a. X-rays removed electrons from fine oil droplets to produce cations b. The rate at which charged oil droplets fell in an adjustable magnetic field was related to the charge of an electron c. The rate at which charged oil droplets fell in an adjustable electric field was related to the charge on an electron d. The rate at which ionized nitrogen and oxygen molecules fell between electrically charged metal plates was related to charge on an electron e. Electrons were found to have either a positive charge or negative charge depending on how they were generated
Answers - General Chemistry I (CHEM 111)
Which of the following statements is true regarding Thomson’s cathode ray experiment and the discovery of electrons? a. Radioactivity was also discovered b. Magnetic fields deflected cathode rays but electric fields did not c. Cathode rays were split into beams by magnetic fields d. The charge of the electron was determined e. The mass-to-charge ratio of the electron was determined
Answers